√70以上 actual yield formula chemistry 271770-What is actual yield in chemistry
Hydrazine, N2H4, is an oily liquid used as a rocket fuel It can be prepared in water by oxidizing ammonia with hypochlorite ions 2 NH3g ClOaq > N2H4aq Claq H2Ol When 350 g of ammonia reacted with an excess of hypochlorite ion, 252 g of hydrazine was produced What is the percentage yield of hydrazine?The percentage yield is the ratio of actual yield to theoretical yield expressed as a percentage (37 g/100 g) × 100% = 37%Y p is the percentage yield (%) Y t is the theoretical yield
/148302528-56a12f323df78cf77268383a.jpg)
Percent Yield Definition And Formula
What is actual yield in chemistry
What is actual yield in chemistry-Percent yield is determined by dividing the actual yield by the theoretical yield and multiplying by 100 Assume that in the cookiebaking example above, you attempted to make 3 dozen (or 36📗 Need help with chemistry?



18 Percentage Yield
Next, identify the decimal percentage yield using the chemical formula The actual yield of acetaminophen was reported as 0198g and is then divided by the theoretical yield of 0217g Notice that the actual yield reported was 019 less than the theoretical yield The decimal percentage of percent yield is 0The Percentage yield is determined by divide the actual yield with the theoretical yield and multiplied by 100% The percent yield equation is given below percent yield = (actual yield/theoretical yield) x 100% Where Actual yield is said to be the amount of product obtained from a chemical reactionMultiply it by 100
In chemistry, yield, also referred to as reaction yield, is a measure of the quantity of moles of a product formed in relation to the reactant consumed, obtained in a chemical reaction, usually expressed as a percentage Yield is one of the primary factors that scientists must consider in organic and inorganic chemical synthesis processes In chemical reaction engineering, "yield", "conversionThe next and final step is plugging the theoretical yield, and the precentage yield (given to us in the original question) into the formula Percentage Yield = (Actual Yield/ Theoretical Yield) * 100 579 = (Actual Yield/100) * 100 Now use algebra 579 = (Actual Yield/100) * 100 100 100 * 579 = (Ay/100) *100 579 = Ay Actual Yield = 579 gramsTheoretical yield is the maximum amount of product that could be received from a chemical reaction under perfect circumstances, but of course chemical reactions are not operating under perfect circumstances What we get instead is the actual yield which is what it's the product that we actually receive from a chemical reaction
Chemistry Is Everywhere Actual Yields in Drug Synthesis and Purification Many drugs are the product of several steps of chemical synthesis Each step typically occurs with less than 100% yield, so the overall percent yield might be very smallA reaction yield is reported as the percentage of the theoretical amount The formula for percentage yield is given by Percentage yield= (Actual yield/theoretical yield )x100 Rearrange the above formula to obtain theoretical yield formula Example 1 Determine the theoretical yield of the formation of geranyl formate from 375 g of geraniolActual yield is the mass of ammonia that is actually produced during the chemical reaction Actual yield of ammonia (NH 3) = 408 g (given in the question) Theoretical yield of ammonia (NH 3) is the mass of product predicted by the balanced chemical equation for the reaction From the
/148302528-56a12f323df78cf77268383a.jpg)


Percent Yield Definition And Formula
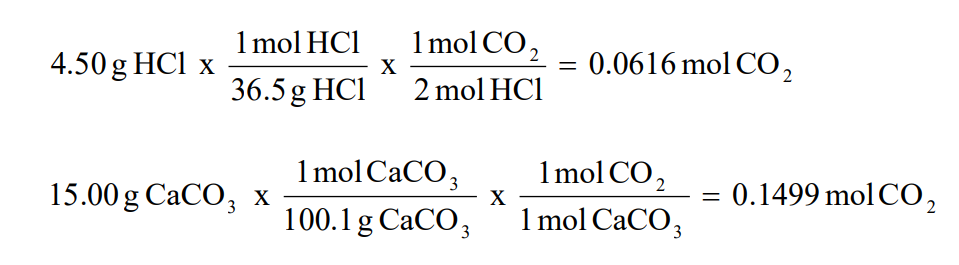


Molecular Formulas And Nomenclature
Theoretical yield is the maximum amount of product that could be received from a chemical reaction under perfect circumstances, but of course chemical reactions are not operating under perfect circumstances What we get instead is the actual yield which is what it's the product that we actually receive from a chemical reactionThe actual yield of a product in a chemical reaction should not exceed its theoretical yield True contains the coefficient 4 in front of the formula Mg3(PO4)2 This term represents that there are 12 Mg atoms, 4 P atoms and 16 O atoms in this term False The mass of one ethanol (C2H6O) molecule is 765 x 10^23 grams trueLearn how to identify the limiting reactant in a chemical reaction and use this information to calculate the theoretical and percent yields for the reaction


Calculation Of Theoretical Yield Organic Chemistry I 212 01



Howto How To Find Percent Yield Without Actual Yield
Actual yield is the mass of ammonia that is actually produced during the chemical reaction Actual yield of ammonia (NH 3) = 408 g (given in the question) Theoretical yield of ammonia (NH 3) is the mass of product predicted by the balanced chemical equation for the reaction From theTheoretical yield formula Using the theoretical yield equation helps you in finding the theoretical yield from the mole of the limiting reagent, assuming 100% efficiency So, to stop you from wondering how to find theoretical yield, here is the theoretical yield formula mass of product = molecular weight of product * (moles of limiting reagent in reaction * stoichiometry of product)The ratio of actual yield to theoretical yield expressed in percentage is called the percentage yield \(\mathrm{percent\ yield = \dfrac{actual\ yield}{theoretical\ yield}\times100}\) Chemical reaction equations give the ideal stoichiometric relationship among reactants and products Thus, the theoretical yield can be calculated from



How To Calculate Percent Yield In Chemistry 15 Steps



Yield Amount Of Products Ppt Download
Theoretical Yield Formula Questions 1 Determine the theoretical yield of H 2 O (in moles) in the following reaction, if 25 moles of hydrogen peroxide are decomposed 2H 2 O 2 → 2H 2 O O 2 Answer In this reaction there is only one reactant (H 2 O 2) so it must be the limiting reactantStoichiometry will be used to determine the moles of water that can be formedLearn how to identify the limiting reactant in a chemical reaction and use this information to calculate the theoretical and percent yields for the reactionThe actual yield of a product in a chemical reaction should not exceed its theoretical yield True contains the coefficient 4 in front of the formula Mg3(PO4)2 This term represents that there are 12 Mg atoms, 4 P atoms and 16 O atoms in this term False The mass of one ethanol (C2H6O) molecule is 765 x 10^23 grams true



Reaction Percent Yield Introduction And Practice Exercises
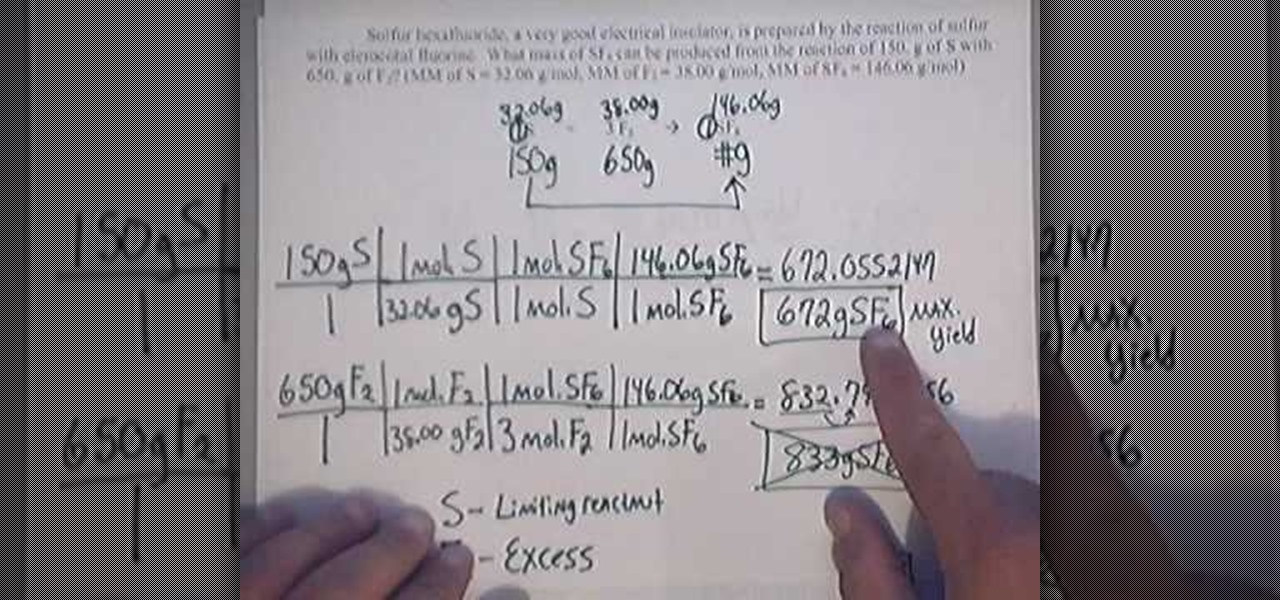


Howto How To Find Percent Yield Without Actual Yield
The actual yield of 37 g is the amount of product that is weighed at the end of a reaction;Actual yield / Theoretical yield X 100% = Percent Yield Why are actual yields generally less than those calculated theoretically?Theoretical yield calculator is an online tool that calculates the theoretical yield in chemical reactions It takes actual yield and percent yield, and finds the amount of yield in a theoretical chemical reaction What is theoretical yield?



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



Calculating Percent Yield Hey Chemistry
In chemistry, actual yield is the amount of substance obtained from the chemical reaction Calculating actual yield is very important to know the reaction efficiency of the reactions in the chemistry labs We can calculate actual yield when we know the percent yield and theoretical yield Formula to calculate actual yield is given by Enter theFormula You can also find the theoretical yield yourself using the theoretical yield formula if percentage yield or actual yield is known Theoretical yield = Actual yield 100 Here is the method Convert both known values into the same unit;Percent yield or percentage yield is the ratio of the actual yield and the theoretical yield of a chemical reaction The experimental yield is divided by the theoretical yield and multiplied by 100 to be calculated as the percent yield If the theoretical yield and the experimental yield are same then the percent yield will be 100%



Solved N Ho What Is The Chemical Formula For The Limiting Chegg Com



Reaction Yield Theoretical Vs Actual Yield Theoretical Yield Is The Maximum Amount Of Product That Can Be Produced From A Given Amount Of Reactant Calculated Ppt Download
An actual yield is the mass of a product actually obtained from the reaction It is usually less than the theoretical yield It is usually less than the theoretical yield The reasons for thisThe formula for calculating the percent yield is Percentage yield = mass of actual yield ÷ mass of theoretical yield × 100% Let's assume that you obtained an actual yield of 850 grams Then, the percent yield would be Percentage yield of NaCl = 850 grams ÷ 993 grams × 100% Percentage yield of NaCl = 8559% Since the value ofSo, ideally, 336 grams of CaO should have been produced in this reaction This is the theoretical yield However, the problem tells us that only 15 grams were produced 15 grams is the actual yield It is now a simple matter to find percent yield



How To Calculate Theoretical Yield And Percent Yield Youtube



Bundle Of Chemistry Step By Step Templates By Effortless Tpt
Full set of chemistry calculators Calculate Actual vs Theoretical Yield for an experiment or process Estimate theoretical yield based on mole ratios Calculate the molar mass of a molecule from the formula Your one stop shop for chemistry calculators and manufacturing calculatorsNext, identify the decimal percentage yield using the chemical formula The actual yield of acetaminophen was reported as 0198g and is then divided by the theoretical yield of 0217g Notice that the actual yield reported was 019 less than the theoretical yield The decimal percentage of percent yield is 0Actual Yield Formula The following is the formula for calculating the actual yield of a process Y a = Y p / 100 * Y t Where Y a us the actual yield;



Percent Yield Calculator



Quantitative Chemistry Theoretical And Percent Yield
The theoretical yield of 100 g is the calculated amount of product, assuming that the reaction is 100% efficient;Next, identify the decimal percentage yield using the chemical formula The actual yield of acetaminophen was reported as 0198g and is then divided by the theoretical yield of 0217g Notice that the actual yield reported was 019 less than the theoretical yield The decimal percentage of percent yield is 0Download 12 Secrets to Acing Chemistry at http//conquerchemistrycom/chemsecrets/💯 If you like my teaching style and are inte



Question Video Calculating The Percentage Yield Of The Recreation Of Aqueous Copper Sulfate With Zinc Metal Nagwa



Unit 8 Percent Yield Calculations Ppt Download
Actual Yield Definition The actual yield is the quantity of a product that is obtained from a chemical reactionIn contrast, the calculated or theoretical yield is the amount of product that could be obtained from a reaction if all of the reactant converted to product Theoretical yield is based on the limiting reactantIf the actual and theoretical yield are the same, the percent yield is 100% Usually, percent yield is lower than 100% because the actual yield is often less than the theoretical value Reasons for this can include incomplete or competing reactions and loss of sample during recoveryDivide actual yield to percent yield;



Percent Yield Made Easy Stoichiometry Tutorial Part 4 Youtube



Howto How To Find Percent Yield Without Actual Yield
The actual yield is the actual amount of product that is produced in a chemical reaction The theoretical yield refers to the amount that should be form when the limiting reagent is completely consumed The actual yield is expressed as a percentage of the theoretical yield This is called the percent yield To find the actual yield, simply multiply the percentage and theoretical yield togetherTheoretical yield formula Using the theoretical yield equation helps you in finding the theoretical yield from the mole of the limiting reagent, assuming 100% efficiency So, to stop you from wondering how to find theoretical yield, here is the theoretical yield formula mass of product = molecular weight of product * (moles of limiting reagent in reaction * stoichiometry of product)An actual yield is, well, the amount of product actually produced by the reaction in a lab or as told to you in the chemistry problem A theoretical yield is the amount of product that could've been produced had everything gone perfectly, as described by theory if every single atom of reactants worked together perfectly



Calculating Actual Yield Given The Percent Yield Youtube



Stoichiometry And Percent Yield Examples Solutions Worksheets Videos Games Activities
Theoretical Yield Formula Questions 1 Determine the theoretical yield of H 2 O (in moles) in the following reaction, if 25 moles of hydrogen peroxide are decomposed 2H 2 O 2 → 2H 2 O O 2 Answer In this reaction there is only one reactant (H 2 O 2) so it must be the limiting reactantStoichiometry will be used to determine the moles of water that can be formedIn chemistry, actual yield is the amount of substance obtained from the chemical reaction Calculating actual yield is very important to know the reaction efficiency of the reactions in the chemistry labs We can calculate actual yield when we know the percent yield and theoretical yield Formula to calculate actual yield is given by Enter theAn actual yield is the mass of a product actually obtained from the reaction It is usually less than the theoretical yield It is usually less than the theoretical yield The reasons for this



Actual Yield Amount Of Product That Is Actually Produced When The Chemical Course Hero



Limiting Reactant And Reaction Yields Article Khan Academy
The theoretical yield is a term used in chemistry to describe the maximum amount of product that you expect a chemical reaction could create You need to begin with a balanced chemical equation and define the limiting reactant When you measure the amount of that reactant that you will be using, you can calculate the amount of productIn chemistry, the theoretical yield is the maximum amount of product a chemical reaction could create based on chemical equationsIn reality, most reactions are not perfectly efficient If you perform the experiment, you'll end up with a smaller amount, the actual yieldTo express the efficiency of a reaction, you can calculate the percent yield using this formula %yield = (actual yield📗 Need help with chemistry?



Percent Yield Calculator 100 Free Calculators Io



Reaction Yield Protocol
Download 12 Secrets to Acing Chemistry at http//conquerchemistrycom/chemsecrets/💯 If you like my teaching style and are intePercent yield or percentage yield is the ratio of the actual yield and the theoretical yield of a chemical reaction The experimental yield is divided by the theoretical yield and multiplied by 100 to be calculated as the percent yield If the theoretical yield and the experimental yield are same then the percent yield will be 100%Theoretical, actual and percentage yield Theoretical yield the maximum possible mass of a product that a chemical reaction can make It is calculated using molar ratios



How To Calculate Percent Yield In Chemistry 15 Steps



How To Calculate Percent Yield 3 Ways To Solve Chemistry Problems Tripboba Com
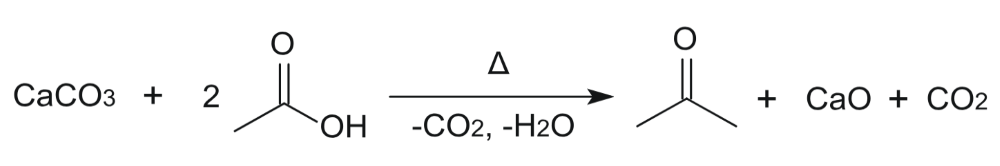
Theoretical yield can be defined as quantity of a product acquired from complete conversion of the limiting reactant in a chemical reactionReactants often yield quantities of products that are less than those calculated based on the formulated chemical reaction The per cent yield formula is used to determine the percentage of the theoretical yield that was formed in a reactionLess than complete reactions, impure reactants, and reactants left in the beakers



Reaction Percent Yield Introduction And Practice Exercises



How To Calculate Percent Yield In Chemistry 15 Steps



Percent Yield Calculator


Percent Yield Calculator



Percent Yield Chemistry Video Clutch Prep
/pipetting-sodium-carbonate-to-copper--ii--sulfate--both-solutions-0-5-m-concentration--copper--ii--carbonate-precipitate-formed-result--cuso4---na2co3----cuco3---na2so4--double-displacement-reaction-565785491-59232e6f3df78cf5fa2d92e3.jpg)


Definition Of Theoretical Yield In Chemistry



Percentage Yield Lab Answers Schoolworkhelper



How To Calculate Percentage Yield In Chemistry How To Wiki



Calculating Percent Yield Hey Chemistry



How To Calculate Theoretical Yield 12 Steps With Pictures



Percent Yield Percent Purity Solutions Examples Videos



Document



Image Result For Theoretical Yield School Work Chemistry Basic Concepts



Reaction Percent Yield Introduction And Practice Exercises


8 6 Limiting Reactant Theoretical Yield And Percent Yield From Initial Masses Of Reactants Chemistry Libretexts


Www Assignmentexpert Com Homework Answers Chemistry Answer Pdf



Theoretical Yield Vs Actual Yield Sample Lab Data
/chemical-reaction-pipette-0-25-m-solution-of-iron-iii--chloride--fecl3--into-0-5-m-solution-of-sodium-carbonate--na2co3---rusty-red-iron-iii--hydroxide-precipitate--fe-oh-3----carbon-dioxide-produced-667616691-5921d7385f9b58f4c086581b.jpg)


Actual Yield Definition Chemistry



How To Find Actual Yield Theoretical Yield And Percent Yield Examples Practice Problems Youtube



Theoretical Actual And Percent Yield Problems Chemistry Tutorial Youtube


Calculating Percentage Yield



How To Calculate Percent Yield In Chemistry 15 Steps



How To Calculate And Solve For Percentage Yield Actual Yield And Theoretical Yield In Chemistry The Calculator Encyclopedia Nickzom Blog



Yield Calculations Faculty Staff Sites



Solved During A Chemical Reaction When Two Substances Rea Chegg Com



Limiting Reactants Unit



Howto How To Find Percent Yield Without Actual Yield


How To Calculate And Solve For Percentage Yield Actual Yield And Theoretical Yield In Chemistry The Calculator Encyclopedia Nickzom Blog



Limiting Reactant And Percent Yield


Yield Calc



Percentage Yield Lab Answers Schoolworkhelper
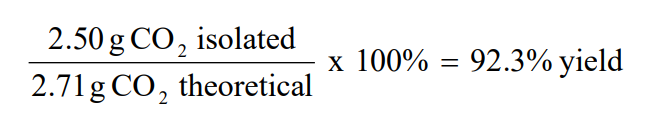


Molecular Formulas And Nomenclature



How To Calculate The Percent Yield And Theoretical Yield Youtube


Q Tbn And9gctx2unihrwvsl45ij5h Bp2grxrgaokdparonvakfmrfmh9cyz4 Usqp Cau



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com


The Stoichiometry Of Product Formation And Percent Yield



How To Calculate Percent Yield In Chemistry 15 Steps



Percent Yield And Percent Error Calculations Science Chemistry Percent Yield Percent Error Showme



18 Percentage Yield



3 4 Percent Yield Chemistry Libretexts



How To Calculate And Solve For Percentage Yield Actual Yield And Theoretical Yield In Chemistry The Calculator Encyclopedia Nickz Chemistry Solving Actual



Theoretical Yield Calculator Learn Reaction S Efficiency



Ch 3 2 Formula Moles And Equations



Calculating Reaction Yield And Percentage Yield From A Limiting Reactant Science Class Video Study Com


Q Tbn And9gcrynatxciaobbvt Edbzcqmlgnhpv Acw5cqav2pwzm1fudi Ih Usqp Cau



Percentage Yield Lab Answers Schoolworkhelper


Www Usna Edu Chemdept Files Documents 111pdf Ay 21 Documents Ch 7 Sc111 Learning Objectives Ay21 Pdf



Yields Introductory Chemistry



C Calculate Your Actual Yield Of Sodium Acetate Chegg Com



How To Calculate Percent Yield In Chemistry 15 Steps


4 4 Reaction Yields Chemistry



Unit 8 Percent Yield Calculations Ppt Download


Calculation Of Theoretical Yield Organic Chemistry I 212 01



Theoretical Actual Percent Yield Error Limiting Reagent And Excess Reactant That Remains Youtube



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



How To Calculate Percentage Yield In Chemistry How To Wiki



Jobian Rijo Percentage Yield Doc Chemistry Journal 5 06 Percent Yield Driving Question How Do Chemists Use Percentages And Stoichiometry To Understand Course Hero



Oneclass Limiting Reagent And Percent Yield Practice In A Chemical Reaction With More Than One React



Percent Yield Concept Chemistry Video By Brightstorm



Lesson Percentage Yield



Nwtc General Chemistry Ch 09



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com


Q Tbn And9gcs60s5akezigys4bjyqzve5tdrkwvatrchodzxvxw7 9iinr194 Usqp Cau



Calculating Percent Recovery Percent Yield Chemical Reactions Unit Processes



Reaction Yield Protocol



Percent Yield Tutorial Explained Practice Problems Crash Chemistry Academy Youtube



Howto How To Find Percent Yield Without Actual Yield
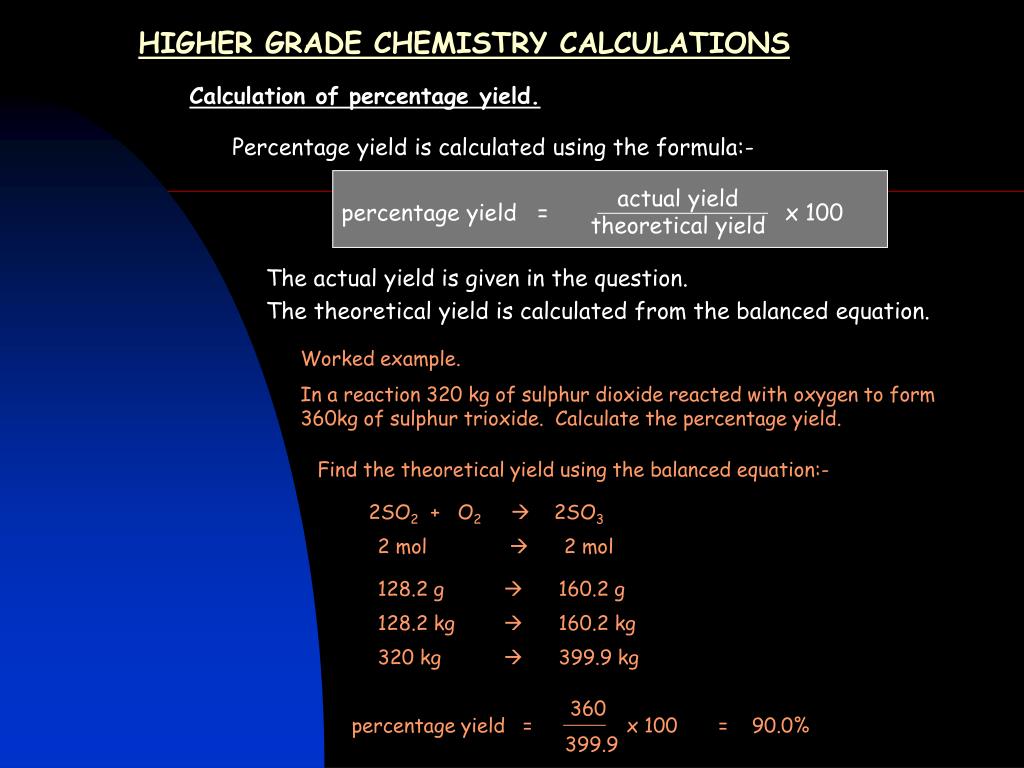


Ppt Higher Grade Chemistry Calculations Powerpoint Presentation Free Download Id



Yields Introductory Chemistry



Stoichiometry Solving Percent Yield Stoichiometry Problems Youtube



Howto How To Find Percent Yield Without Actual Yield



Solved Limiting Reactant And Percent Yield Lab Nameeuion Chegg Com



Chemistry 101 Calculating Reactant Given Percent Yield And Actual Product Youtube



Percent Yield Percent Purity Solutions Examples Videos


Q Tbn And9gcsoowhfh Ellws R7fhtegyfftxnyz 7tf9b1pxdba108ocwhdh Usqp Cau


コメント
コメントを投稿